 German researchers at Technische Universität München (TUM) have studied how electronic interactions govern the encounter between a molecule called porphine and copper and silver surfaces.
German researchers at Technische Universität München (TUM) have studied how electronic interactions govern the encounter between a molecule called porphine and copper and silver surfaces.
As electronic components continue to shrink, top-down manufacturing methods begin to approach a physical limit at the nanoscale. Rather than continue to chip away at this limit, one solution involves using the bottom-up self-assembly of molecular building blocks to build nanoscale devices.
For successful self-assembly to work the attractive and repulsive forces within molecules, between each molecule and its neighbours, and between molecules and the surface that supports them, all have to be taken into account. To better understand the self-assembly process, researchers at TUM have characterised the contributions of all interaction components, such as covalent bonding and van der Waals interactions between molecules and between molecules and a surface.
"In an ideal case, the smallest possible device has the size of a single atom or molecule," said Katharina Diller, who worked as a postdoctoral researcher at TUM.
One such example is a single-porphyrin switch, which occupies a surface area of 1nm2. The porphine molecule, which was the object of this study, is even smaller than this. Porphyrins are a group of ringed chemical compounds which include haem. Haems are most commonly recognised as components of haemoglobin, responsible for transporting oxygen and carbon dioxide in the bloodstream and chlorophyll in plants. In synthetically-derived applications, porphyrins are studied for their potential uses as sensors, light-sensitive dyes in organic solar cells, and molecular magnets.
The researchers from TUM assessed the interactions of the porphyrin molecule 2H-porphine by using density functional theory, a quantum mechanical computational modelling method used to describe the electronic properties of molecules and materials. Their simulations were performed on SuperMUC, the high-performance supercomputer at Leibniz-Rechenzentrum.
The single crystal surfaces of copper and silver metallic substrates the researchers chose for the porphyrin molecules to assemble on are widely used as substrates in surface science. This is due to the densely packed nature of the surfaces, which allow the molecules to exhibit a smooth adsorption environment. Additionally, copper and silver each react differently with porhyrins - the molecule adsorbs more strongly on copper, whereas silver does a better job of keeping the electronic structure of the molecule intact, allowing the researchers to monitor a variety of competing effects for future applications.
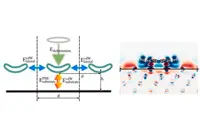
In their simulation, porphyrin molecules were placed on a copper or silver slab. After finding the optimal geometry in which the molecules would adsorb on the surface. To simulate different molecular coverages, the researchers altered the size of the metal slab to increase or decrease the distance between molecules. The computational setup gave them a switch to turn the energy contributions of neighbouring molecules on and off, in order to observe the interplay of the individual interactions.
The team found that the weak long-range van der Waals interactions yielded the largest contribution to the molecule-surface interaction, and showed that the often employed methods to quantify the electronic charges in the system have to be used with caution. While interactions directly between molecules are negligible, the researchers said they found indications for surface-mediated molecule-to-molecule interactions at higher molecular coverages.
"The analysis of the electronic structure and the individual interaction components allows us to better understand the self-assembly of porphine adsorbed on copper and silver, and additionally enables predictions for more complex porphyrine analogues," Diller said. "These conclusions, however, come without yet considering the effects of atomic motion at finite temperature, which we did not study in this work."
Pic: Schematic depiction of different energy terms contributing to the adsorption energy, and charge density difference of 2H-P after adsorption onto Cu(111) at 12.8 Angstrom separation
Author
Tom Austin-Morgan
Source: www.newelectronics.co.uk

